Which Terms Are Used to Describe Pure Substances
The terms homogeneous and heterogeneous are not usually used to describe pure substances. Physical properties can be used to identify a pure substance.
What Is A Pure Substance In Thermodynamics Quora
Physical properties of a substance are characteristics that can be observed without altering the identity of the substance.
. Compounds are pure substances. A pure substance participates in a chemical reaction to form predictable products. A pure substance does not have to be of a single element or compound.
Color odor density melting temperature boiling temperature and solubility are examples of physical properties. The substances that are free from any kind of mixture and contain only one kind of particle are pure substances. Separation by a physical process.
The agate stone cannot be a compound because materials with different properties appear as irregular rings in the stone. Examples of mixture include the salt solution which is a mixture. The type of atoms in a pure substance determine if the substance is compound or an element.
33 use the particle theory to describe the difference between pure substances which have identical particles and mixtures which have different particles 34 distinguish between solutions and mechanical mixtures 35 describe the processes eg evaporation sifting filtration distillation magnetism used to. The Particle Theory Of Pure Substances And Mixtures. Used to describe a substance that is not uniform in composition.
If there are two kinds of atom in a molecule it is a compound. Physical properties can be. If there is only one kind of atom in a molecule or crystal matrix like a metal it is an element.
Some examples of pure substance that are elements are sulfur and tin. B Boiling point of water decreases with increasing pressure. A Specific volume of water decreases on freezing.
The particle Theory explains that both Pure substances and Mixtures are made up of particles which are in continuous motion. Is a mixture that looks like a single pure substance - a uniform mixture of two or more pure substances. Pure Substance A substance that has a fixed chemical composition throughout is called a pure substance such as water air and nitrogen.
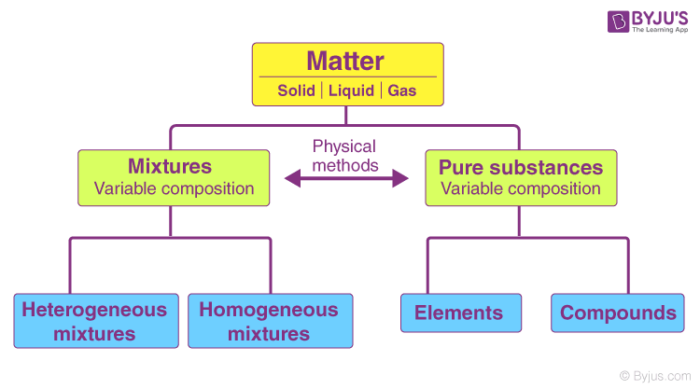
The terms element and compound are used to identify pure substances. A mixture in which different materials can be identified easily homogeneous mixture. Substances that have two or more different particles are mixtures.
Used to describe a substance that is uniform in composition or character. What terms are used to identify pure substances. They have constant composition and properties throughout.
Color odor density melting temperature boiling temperature and solubility are examples of physical properties. For example salt is a pure substance made only of sodium chloride. The substances that are made of a single element or compound are called pure substances.
A pure substance or chemical substance is a material that has a constant composition is homogeneous and has consistent properties throughout the sample. What is the term used to describe substances that cannot be decomposed into two or more simpler substances by chemical means. If there are two kinds of atoms in a crystal metal matrix it is a mixture and not a pure substance.
D Freezing temperature of water decreases with increasing pressure. Thermodynamics Properties of Pure Substances 1. - Pure Chemical Substance - Compound All compounds are pure.
These Particles can be the same and also different. The purity of a substance is the measure of the extent to which a given substance is pure. If this sample of sodium chloride NaClNaCl had been impure we would call it a mixture not a compound.
Pure substances are defined as substances that are made of only one type of atom or molecule. In chemistry a pure substance consists of only one type of atom molecule or compound. A substance with two or more elements that are combined inthe same proportion element.
A mixture that contains two or more substances blended evenly throughout. These can be split into two types. Which terms are used to identify pure substances.
Also known as a homogeneous mixture. Each of these stats of matter Can be a pure substance and a mixture For. A substance inwhich allthe atoms are the same heterogeneous mixture.
Physical properties of a substance are characteristics that can be observed without altering the identity of the substance. A mixture that looks like a single pure substance. A uniform mixture of two or more pure substances.
This means that solid liquids And gasses Are made up of particles. Several elements compounds together form a mixture. Substances are made from only one chemical or one.
A mixture of two or more phases of a pure substance is still a pure substance as long as the chemical. A mixture of water and salt is an example of a homogeneous mixture because the salt is evenly mixed. Solutions and mechanical mixtures.
Examples of pure substances include iron aluminum silver and gold. Pure substance can be identified as either elements or compounds. This term can be used to describe all pure substances and solutions.

What Are The Types Of Pure Substances Compounds Elements Videos

Pure Substances And Mixtures Venn Diagram Examples Venn Diagram Worksheet Venn Diagram Template

What Is Pure Substance Definition Examples Difference Between Pure Substance Mixture
Comments
Post a Comment